Canine transmissible venereal tumour (CTVT) was the
first naturally occuring neoplasm that is spread by the transferring
of cells from an affected dog to another. It happens to be venereally
transmitted. The second neoplasm to be spread in a similar fashion
is the oral facial tumour of Tasmanian devils, called Devil Facial
Tumour Disease. The tumours are therefore allographs. While this has been
known for many years, it has been definitively established by recent
molecular techniques. There are several reviews written about the
CTVT (Cohan 1978, Mukaratirwa and Gruys 2003, Mello-Martins et al
2005)
The cells of CTVT have a chromosomal number of 57,
58 or 59. The dog has 78. Weber et al (1965) examined the chromosomes
of CTVT in the USA, and they were nearly identical to those reported
in Japan. They suggested that the CTVT was a transplanted tumour
that arose from one dog. Dogs with the neoplasm spread it to other
dogs, so the tumour has always been regarded as an infectous tumour.
More recently, Murgia et al (2006) used molecular techniques to
identify that neoplasms from different continents and collected
decades apart are clonal, and, while there are 2 subtypes, they
have a common origin. The DNA of the CTVT has closely related DNA
of wolves and East Asian dog breeds.
The cells of a CTVT are able to avoid detection by
the immune cells. They downregulate class 1 molecules and there
is no class 2 activity, because they secrete inhibitory cytokines
(TGFbeta1 and IL 6) (Liao et al 2003, Murgia et al 2006). In the
initial proliferative phase of CTVT, they express little MHC Class
1 or 2. After about 12 weeks in an experimental model, MHC expression
increased dramatically and was associated with the presence of lymphocytes;
and at the same time, the masses stopped growing. It appeared that
the lymphocytes stimulated MHC expression and are responsible for
regression of the tumours (Hsiao et al 2002).
CTVT are usually found on the penis, but they are
occasionally seen in the mouth (Bright et al 1983), and metastasis
occurs to the skin and throughout the body, including the brain (subdural and extraaxial). There is one case of ocular CTVT. This occurs especially
in dogs that are immunosuppressed with whole body irradiation. It
is a rare occurrence naturally. One case of metastasis was found in the brain.
The phenotype of the cells have been the subject of
considerable debate over the years. They have features and staining
characteristics that suggest they are histiocytic in origin (see
below).
Dogs usually respond to treatment with vincristine. In one study of 38
dogs with CTVT, 31 responded to vincristine. One dog died, and 6 required
treatment with doxorubricin. There were no reoccurrences. (Nak et al 2005)
CTVT are also known to regress spontaneously, and there is evidence that host immunity is important in this and in progression. Paia et al (2011) enhansed regression by fusing allogenic dendritic cells with neoplastic cells and using them as a vaccine in affected dogs. Vaccinated dogs has reduced progression and enhanced regression over unvaccinated controls.
Stockman et al (2011) examined CTVT for the proteins p53, p63 and Bcl2, and found that all were present at all stages of the disease. These proteins are involved with induction of apoptosis.
Marino et al (2012) identified that dogs with leishmaniasis frequently had Leishmania organisms in macrophages in their tumours.
Murchison et al (2014) provides a state of the art publication on the genetics of CTVT.
Extragenital CTVT is more frequently seen in male dogs and in an oronasal locaton (
Macroscopic findings
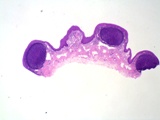
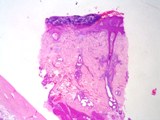
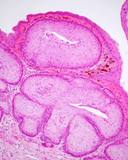
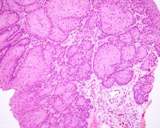
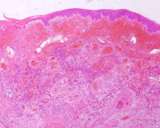
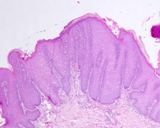
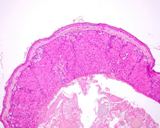
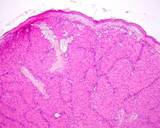
The lesions of transmissible venereal tumours are exophytic multinodular
proliferations in the preputial cavity, often attached to the junction
between the inner sheath of the prepuce and penile epithelium. The size
can vary considerably from small nodules to large fungating masses that
cause preputial swellings.

Figure : Transmissible venereal tumour, penis and prepuce
Microscopic findings
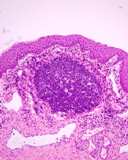
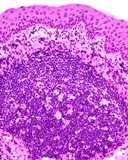
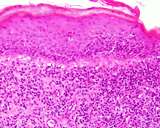
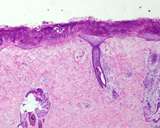
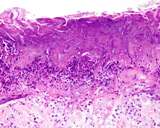
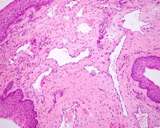
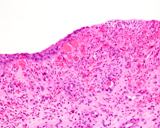
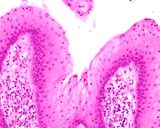
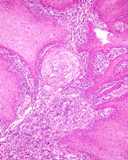
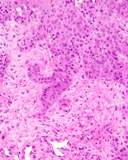
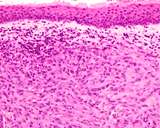
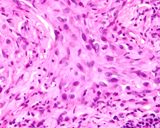
Transmissible venereal tumours are round cell tumours. They are locally
infiltrative, and the cells are often arranged in packets with a fine
stroma. The cells have well defined cytoplasmic boundaries, abundant often
clear cytoplasm and a central round to indented nucleus. Mitoses are numerous.
Some have fine vacuoles around the cytoplasmic membrane , and this feature
is particulary prominent in cytological specimens. Cells such as macrophages
and lymphocytes may be found in the neoplasms.
The differential diagnoses include lymphoma, histiocytic tumour, mast
cell tumour and plasmacytoma.
Immunohistochemistry
Sandusky et al (1997) stained 4 CTVT with S-100, kappa and lambda light
chains, alpha-1-antitrypsin, alpha-1-antichymotrypsin, LCA, neuron-specific
enolase, keratin, cytokeratin, muramidase, and vimentin. The CTVT were
consistently vimentin positive
Mozos et al (1996) reported on 25 CTVT stained for human lysozyme, human
alpha-1-antitrypsin, CD3, vimentin, human keratins, human lambda light
chains, canine immunoglobulins IgG, IgM, and S-100. Lysozyme was immunoreactive
in 10/25 alpha-1-antitrypsin in 14/25, and vimentin in 25/25 CTVT. All
were negative to keratins, S-100, lambda light-chain, IgG, IgM, and CD3.
Marchal et al (1997) reported CTVT stained for "lysozyme, ACM1 antigen,
vimentin, neuron-specific enolase, glial fibrillary acidic protein, desmin,
alpha smooth muscle actin, CD3, IgG, kappa and lambda light chains, and
keratin. Lysozyme immunoreactivity was detected in all cases, ACM1 antigen
in 11 of 14, neuron-specific enolase in 11 of 14, vimentin in 10 of 14,
glial fibrillary acidic protein in 4 of 14 and desmin in 1 of 14. All
the sections were negative to keratins, alpha smooth muscle actin and
CD3, whereas in five cases, perivascular tumour cells contained Ig G,
kappa and lambda light chains".
In summary then, CTVT are consistently vimentin positive, cytokeratin
and S100 negative round cell tumours that do not stain for specific T
or B lymphocyte markers and which stain for histiocytic markers.
Bright RM. Gorman NT, Probst CW, Goring CW. (1983) Transmissible venereal
tumor of the soft palate in a dog. J Amer Vet Med Assoc 183: 893-894.
Cohen D (1973) The biological behaviour of the transmissible venereal
tumour in immunosuppressed dogs. Europ J Cancer 9: 253-258
Cohen D (1978) The transmissible venereal tumor of the dog - a naturally
occurring allograft? a review. Israel J Med Sc 14: 14-19.
Dass LL, Sahay PN (1989) Surgical treatment of canine transmissible venereal
tumour - a retrospective study. Indian Vet J 66: 255-258.
Ganguly B, Das U, Das AK. Canine transmissible venereal tumour: a review. Vet Comp Oncol. 2016; 14: 1-12
Higgins DA (1966) Observations on the canine transmissible Venereal Tumour
as seen in the Bahamas. Vet Rec 79: 67-71.
Hsiao YW, Liao KW, Hung SW, Chu RM (2002) Effect of tumor
infiltrating lymphocytes on the expression of MHC molecules in canine
transmissible venereal tumor cells. Vet Immunol Immunopathol. 87(1-2):19-27.
R. M. Kabuusu, D. F. Stroup, C. Fernandez (2010) Risk factors and characteristics
of canine transmissible venereal tumours in Grenada, West Indies. Vet
Comp Oncology 8: 50-55.
Liao KW, Hung SW, Hsiao YW, Bennett M, Chu RM (2003) Canine transmissible
venereal tumor cell depletion of B lymphocytes: molecule(s) specifically
toxic for B cells. Vet Immunol Immunopathol 92(3-4):149-162.
Marchal T, Chabanne L, Kaplanski C, Rigal D, Magnol JP.(1997) Immunophenotype
of the canine transmissible venereal tumour. Vet Immunol Immunopathol.
57(1-2):1-11.
Marino G, Gaglio G, Zanghì A. (2012) Clinicopathological study of canine
transmissible venereal tumour in leishmaniotic dogs. J Sm An Pract 2012,
Mello-Martins MI, Ferreira de Souza F, Gobello C (2005) Canine Transmissible
Venereal tumor: etiology, pathology, diagnosis and treatment. In: Recent
Advances in Small Animal Reproduction. Concannon PW, England G, Verstgegen
J, Linde-Forsberg (Eds) www.ivis.org.
Milo J, Snead E. A case of ocular canine transmissible venereal tumor.Can Vet J. 2014; 55: 1245-1249.
Mozos E, Mendez A, Gomez-Villamandos JC, Martin De Las Mulas J, Perez
J. (1996) Immunohistochemical characterization of canine transmissible
venereal tumor. Vet Pathol33(3):257-263
Mukaratirwa S, Gruys E. (2003) Canine transmissible venereal tumour:
cytogenetic origin, immunophenotype, and immunobiology. A review. Vet
Q. 25(3):101-111.
Murchison EP, Wedge DC, Alexandrov LB, Fu B, Martincorena I, Ning Z, Tubio JM, Werner EI, Allen J, De Nardi AB, Donelan EM, Marino G, Fassati A, Campbell PJ, Yang F, Burt A, Weiss RA, Stratton MR. Transmissible [corrected] dog cancer genome reveals the origin and history of an ancient cell lineage. Science. 2014; 24: 343, 437-440.
Murgia C, Pritchard JK, Kim SY, Fassati A, Weiss RA. (2006) Clonal origin
and evolution of a transmissible cancer Cell. 126(3):477-487.
Nak D, Nak Y, Cangul IT, Tuna B. (2005) A clinicopathological study on
the effect of vincristine on transmissible venereal tumour in dogs. J
Vet Med 52: 366-370.
Paia Chien-Chun, Kuob Tzong-Fu, Maoc Simon J.T. , Chuanga Tien-Fu, Lina Chen-Si, Chua Rea-Min (2011) Immunopathogenic behaviors of canine transmissible venereal tumor in dogs following an immunotherapy using dendritic/tumor cell hybrid Veterinary Immunology and Immunopathology 2011 139: 187–199
Parent R, Teuscher E, Morin M, Buyschaert A. (1983). Presence of the
canine transmissible venereal tumor in the nasal cavities of dogs in the
area of Dakar (Senegal). Canadian Vet J 24: 287-288.
Rogers KS, Walker MA, Dillon HB (1998) Transmissible venereal tumor:
a retrospective study of 29 cases. J Amer Anim Hosp Assoc 34: 463-470.
Sandusky GE, Carlton WW, Wightman KA. (1987) Diagnostic immunohistochemistry
of canine round cell tumors. Vet Pathol. 24(6):495-499
Stockmann D, Ferrari HF, Andrade AL, Cardoso TC, Luvizotto MCR. (2011) Detection of the tumour suppressor gene TP53 and expression of p53, Bcl-2 and p63 proteins in canine transmissible venereal tumour. Vet Comp Oncol 2011, 9: 251-259
Strakova A, Baez-Ortega A, Wang J, Murchison EP. Sex disparity in oronasal presentations of canine transmissible venereal tumour. Vet Rec. 2022 Jul 3:e1794.
Stockmann D, Ferrari HF, Andrade AL, Cardoso TC, Luvizotto MCR. (2011) Detection of the tumour suppressor gene
TP53 and expression of p53, Bcl-2 and p63 proteins in canine transmissible venereal tumour. Vet Comp Oncol 2011, 9: 251-259
Thornburn MJ, Gwynn RVR, Ragbeer MS, Lee BI. (1968) Pathological and
cytogenetic observations on the naturally occurring canine venereal tumour
in Jamaica (Sticker's tumour). British J Canccer 22: 720-727.
Weber WT, Nowell PC, Hare WCD (1965) Chromosome studies of a transplanted
and primary canine venereal sarcoma. J Nat Cancer Inst 35: 537-547.
Wright DH, Peel S, Cooper EH, Hughes DT. (1970) Transmissible venereal
sarcoma of dogs. A histochemical and chromosomal analysis of tumours in
Uganda. Rev Europ etudes Clin Et Biol 15: 155-160.